Roivant/Immunovant - Batoclimab Phase 3 Results
- rspace2
- Apr 15, 2025
- 7 min read
Updated: Apr 20, 2025
Executive Summary
Immunovant will soon publish their phase 3 results for batoclimab, a monoclonal antibody developed to treat Myasthenia Gravis. The drug already passed P3 trials in China with very similar results to existing drugs on the market. It is improbable the company will be unable to replicated the China results in this trial.
There are a few ways to play this catalyst. First, you can buy shares or calls in Immunovant, which has seen a pretty drastic decline over the recent months. Calls have a relatively high implied volatility but it may be worth trying to leverage this position. The catalyst is due Q1 2025, which likely means March, so I would be looking at the April calls.
Another way to play this is buying shares or calls in Roivant Sciences. They own about 60% of the company. The April calls appear relatively cheap, so that may be the cheapest way to play the catalyst if the monkeys running the stock market can figure out Roivant should go up if the drug works.
Background Information
Myasthenia Gravis
Myasthenia gravis (MG) is a chronic autoimmune neuromuscular disorder that leads to muscle weakness and fatigue. It occurs when the body's immune system produces antibodies that interfere with communication between nerves and muscles, specifically at the neuromuscular junction. This prevents proper muscle contraction, leading to weakness.
MG encompasses several subtypes of disease:
1. Generalized Myasthenia Gravis (gMG)
This is the most common form, affecting multiple muscle groups, including those responsible for movement, breathing, and swallowing.
2. Ocular Myasthenia Gravis (oMG)
Affects only the eye muscles, causing ptosis (drooping eyelid) and diplopia (double vision)
Some cases progress to generalized MG, but others remain localized
3. Seropositive vs. Seronegative MG
This classification is based on the presence or absence of autoantibodies in blood tests.
a. Seropositive MG
Patients have detectable antibodies against:
Acetylcholine receptor (AChR) antibodies (most common)
Muscle-specific kinase (MuSK) antibodies (less common but often more severe)
Low-density lipoprotein receptor-related protein 4 (Lrp4) antibodies (rare)
b. Seronegative MG
No detectable AChR, MuSK, or Lrp4 antibodies
Symptoms are similar, but the disease mechanism is less understood
4. Congenital Myasthenic Syndromes (CMS)
Not an autoimmune disorder, but a genetic (inherited) neuromuscular disease
Symptoms appear in infancy or childhood
Caused by mutations affecting neuromuscular transmission
5. Neonatal Myasthenia Gravis
Occurs when a mother with MG passes autoantibodies to her baby during pregnancy
Temporary condition that resolves after birth as the baby clears the antibodies
However, 85% of MG cases are generalized MG with seropositive AChR antibodies. The prevalence of MG is estimated to be about 87 cases per 100,000 people in the US.
The prognosis of MG is generally positive; long-term survival is high and many patients lead nearly normal lives. However, this is highly individual, and some subtypes and some individuals experience a very debilitating disease progression.
Diagnosis is done through a multipart evaluation:
1. Clinical Examination
Muscle Weakness Pattern:
Fluctuating weakness that worsens with use and improves with rest
Commonly affects eyes (ptosis, diplopia), bulbar muscles (speech, swallowing), and limb muscles
2. Antibody Testing (Serological Tests)
Used to detect autoantibodies against neuromuscular junction proteins:
Antibody | Presence in MG Cases | Implications |
AChR (Acetylcholine Receptor) Antibodies | ~85% of generalized MG cases; ~50% of ocular MG | Most common marker |
MuSK (Muscle-Specific Kinase) Antibodies | 5–8% of generalized MG | More severe symptoms |
Lrp4 (Low-Density Lipoprotein Receptor-Related Protein 4) Antibodies | Rare (~2–3%) | Seen in seronegative MG |
Anti-Striational Antibodies | Found in thymoma-associated MG | Can indicate thymic involvement |
3. Electrophysiological Tests
These assess neuromuscular transmission by evaluating nerve-muscle responses:
a. Repetitive Nerve Stimulation (RNS) Test
A nerve is repeatedly stimulated, and muscle response is measured
A characteristic "decrement" (progressive decline in response) is seen in MG
Most sensitive in generalized MG, less so in ocular MG
b. Single Fiber Electromyography (SFEMG)
Most sensitive test for MG
Measures "jitter" (variation in time between nerve impulse and muscle response)
Detects abnormalities even if antibody tests are negative
There can also be imaging studies if a thymoma is suspected or respiratory studies if the MG is severe.
Management
The current management if MG is outlined below:
1. First-Line Symptomatic Treatment
Acetylcholinesterase Inhibitors
Pyridostigmine (Mestinon)
Improves neuromuscular transmission by increasing acetylcholine at the neuromuscular junction
Provides short-term symptom relief, but does not stop disease progression
Most effective in mild to moderate MG
2. Immunomodulatory Therapy (Disease-Modifying Treatments)
For moderate to severe generalized MG, long-term immune suppression is required.
Corticosteroids (First-Line Immune Suppression)
Prednisone or Prednisolone
Rapid improvement but may worsen symptoms initially
Tapered gradually to minimize side effects
Steroid-Sparing Immunosuppressants (Used in Long-Term Management)
Azathioprine (Imuran) – Commonly used, but takes months to take full effect
Mycophenolate mofetil (CellCept) – Fewer side effects than azathioprine
Cyclosporine & Tacrolimus – Used in refractory cases
Methotrexate & Rituximab – Sometimes used in MuSK-positive MG
3. Targeted Biologic Therapies (For Refractory MG)
Recent FDA-approved advanced therapies for MG:
Drug | Mechanism | Indication |
Eculizumab (Soliris, 2017) | Complement inhibitor (prevents AChR destruction) | Refractory AChR-positive MG |
Ravulizumab (Ultomiris, 2022) | Longer-acting complement inhibitor | Refractory AChR-positive MG |
Efgartigimod (Vyvgart, 2021) | FcRn inhibitor (reduces autoantibody levels) | Generalized AChR-positive MG |
Rozanolixizumab (Rystiggo, 2023) | FcRn inhibitor | AChR-positive & MuSK-positive MG |
Zilucoplan (Zilbrysq, 2023) | Self-injectable complement inhibitor | Generalized AChR-positive MG |
Other options are available for crisis management. Surgical options may be present for some patients.
Testing
The most common test used in clinical trials to evaluate the change in disease severity is the Myasthenia Gravis Activities of Daily Living (MG-ADL) scale. The scale ranges from 0 to 24. It is important to note this is patient-assessed.
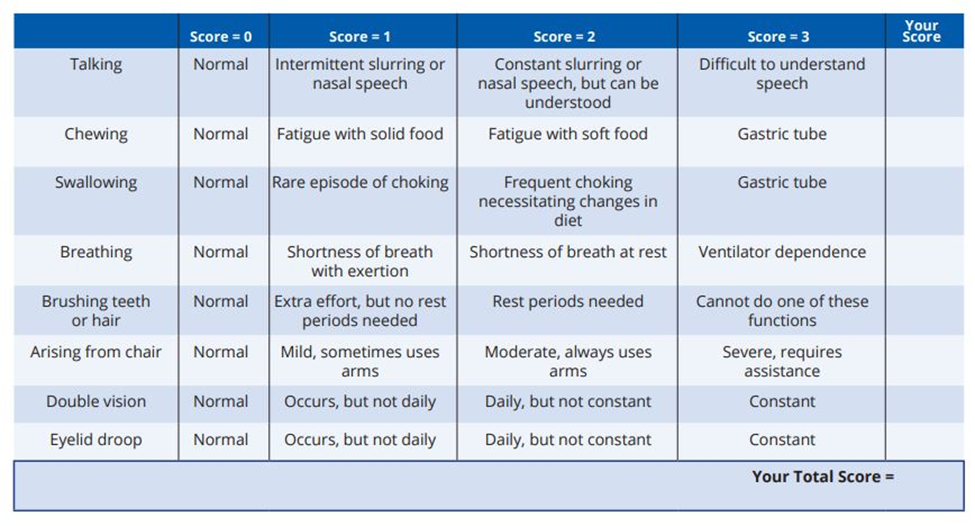
The other standard test commonly used is the Quantitative Myasthenia Gravis (QMG) score.
It evaluates muscle strength across 13 domains, including:
Ptosis (eyelid drooping)
Arm and leg strength
Breathing function
Speech and swallowing
It is clinician-assessed.

Development History
This section is going to focus on the development of the most recent treatments for MG, which are mostly biologics. There are five approved biologics for MG, as outlined in the table above. The following will review the clinical trial results and any post-market information on the approved drugs, focusing on Phase 3 and post-market data.
Soliris – 2017 Approval
a. Did not meet the primary endpoint of improvement in MG-ADL score (p = 0.0698) based on prespecified worst-rank ANCOVA score.
This is a crazy endpoint to use. It makes reaching the endpoint much, much more difficult in a disease where there is always a significant placebo response. It makes it look like the drug does not work when the data does show a clear benefit.
It met the QMG endpoint (p = 0.0129)
This is the money graph from this trial:

2. Ultomiris
To keep things simple, here is the money graph from this trial:

At week 26, there is a significant difference between placebo, which had improved by about 1 point versus the treatment group, which had improved by a little over 3 points in both the QMG and MG-ADL. The open-label extension proved the durability of the effect and the success of switching the placebo group to the treatment group.
3. Vygart

The company decided to use the MG-QOL15 scale instead of the MG-ADL for the visualizations. They did, however, meet the endpoint related to MG-ADL. 67% of treated patients received a clinically significant benefit from the drug, defined as a 2-point or more significant reduction in the MG-ADL score for at least 4 weeks.
4. Rystiggo
“Reductions in MG-ADL score from baseline to day 43 were greater in the rozanolixizumab 7 mg/kg group (least-squares mean change -3·37 [SE 0·49]) and in the rozanolixizumab 10 mg/kg group (-3·40 [0·49]) than with placebo (-0·78 [0·49]; for 7 mg/kg, least-squares mean difference -2·59 [95% CI -4·09 to -1·25], p<0·0001; for 10 mg/kg, -2·62 [-3·99 to -1·16], p<0·0001).”
Sorry, couldn’t pull up a figure for this one. They put the article behind a paywall for some reason, but that line certainly gets the main point across.
5. Zilbrysq
“Patients assigned to zilucoplan showed a greater reduction in MG-ADL score from baseline to week 12, compared with those assigned to placebo (least squares mean change −4·39 [95% CI –5·28 to –3·50] vs −2·30 [–3·17 to –1·43]; least squares mean difference −2·09 [−3·24 to −0·95]; p=0·0004)”
Overall, these drugs appear very similar in efficacy to me.
Batoclimab and Immunovant
Immunovant has an upcoming readout for Batoclimab, a FcRn monoclonal antibody. It has the same MOA as Vygart and Rystiggo. The drug already passed phase 3 trials in China.
“The MG-ADL score diverged between the 2 groups as early as week 2. The mean (SE) maximum difference in MG-ADL score reduction occurred 1 week after the last dose (day 43, 1.7 [0.3] in the placebo group vs 3.6 [0.3] in the batoclimab group; group difference, −1.9; 95% CI, −2.8 to −1.0; nominal P < .001).”
These results appear very similar to all the other phase 3 trials. It would be very improbable for Batoclimab not to meet its endpoint in P3. The only possible scenario would be the data in China was faked. It's unlikely, and I don’t see an obvious incentive for that.
Conclusion
Batoclimab likely works and will meet its primary endpoint in P3. There are a few ways to play this catalyst. First, you can buy shares or calls in Immunovant, which has seen a pretty drastic decline over the recent months. Calls have a relatively high implied volatility but it may be worth trying to leverage this position. The catalyst is due Q1 2025, which likely means March, so I would be looking at the April calls.
Another way to play this is buying shares or calls in Roivant Sciences. They own about 60% of the company. The April calls appear relatively cheap, so that may be the cheapest way to play the catalyst if the monkeys running the stock market can figure out Roivant should go up if the drug works.


Comments